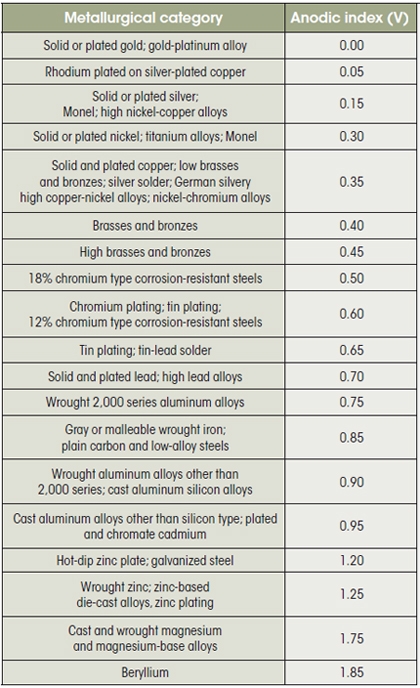
Metals contain thermodynamic energy—positive and negative charges—that are released by the process of corrosion. Metals are rated in their ability to resist electrochemical corrosion on the scale of nobility and on the galvanic series chart, which shows their electrical potential in seawater. More noble metals (such as gold, silver, and titanium) have a neutral or negative electrical potential, so they will not generate a flow of positive ions. At the other end of the scale, more active metals (zinc, aluminum, and cast iron) exhibit strong positive charges that tend to generate electrical current.
Galvanism occurs when two metals with different electrical potential are in close contact, creating a current flow. The resultant flow is very small in dry conditions, but when the metals are joined by water, the water acts as an electrolyte. This causes a significant increase in electron flow from the more-active to the less-active metal, resulting in metallic particles being deposited from the less-- to the more noble metal.
Image

When iron and brass are in close contact with each other in a water-based solution, corrosion will occur. When the iron corrodes, it leaves large pitted, crumbling areas where the material has been removed, which can be seen easily. The brass material shows clearly visible signs of iron oxide (rust) growth. Brass contains no iron and cannot produce iron oxide on its own. Galvanic corrosion must occur for the brass to have iron oxide
attached to its surface.
In the particular instance above, the removal of the cast-iron material directly under the brass wear plate has caused the brass wear plate to break away, owing to a lack of supporting material when under mechanical stresses. The relatively large pieces of brass were found mixed in a fine iron oxide sludge in the bottom of the pump housing, as would be expected for this type of corrosion.
If this damage had been caused by cavitation or fluid contamination instead, the brass material would be attacked at the surface and exposed to pressure or contamination instead of the underlying iron.
THE ELECTRICAL CONNECTION
Image

Corrosion can also occur when dissimilar metals are not in physical contact but are connected electrically by stray electrical currents transferred via a conductive solution, such as contaminated water. When a conductive solution is involved, the process is known as electrolysis. Because the metals are not actually in physical contact, electrolysis can be difficult to trace.
Stray currents energize all underwater metals. The higher electrical potential seeks a path to ground through the weakest metals, starting with the least noble metal, zinc, and moving up the scale of nobility. The culprit is mainly direct current because alternating current needs to be converted into DC before it can cause metallic corrosion. Electrolysis can accelerate the process of corrosion exponentially compared to galvanic reaction alone. The rate of metal deposition will vary depending on the voltage and current applied.
When using a conductive water-based hydraulic fluid, select components carefully. They should use as few dissimilar metals as possible to minimize corrosive potential. Any components used in a water-based hydraulic system should be certified to operate with the chosen fluid by the manufacturer.
INVESTIGATE THE FLUID
Pay close attention to the type of fluid chosen for the system. Some fluids are more conductive (such as water/oil emulsion) than others, creating a high potential for corrosion. Others (such as water glycol) display high electrical resistance, which inhibits corrosion.
Any system using a conductive fluid should be carefully monitored for any stray DC voltage leaks. Just one poor ground connection on equipment using a conductive water-based fluid can cause electrolysis to quickly corrode components, beginning with the least noble metal in the system. Aluminum, cast iron, and steel will typically be the first metals to be attacked. Also, note that every 10° F of temperature rise will double the rate of corrosion.
Image

PREVENTIVE MEASURES
Image
Some precautions can be taken to minimize corrosion due to galvanism and electrolysis:
- Apply coatings to metal surfaces to create a barrier between the metal and the electrolyte solution.
- Incorporate sacrificial materials (such as zinc) into the system to be preferentially corroded instead of allowing important metals to corrode. Unfortunately, this technique introduces a contaminant (zinc oxide) into the fluid.
- Select a fluid with low electrical conductivity.
- Specify components made of materials that are high on the scale of nobility.
Many variables exist in every system, and a perfect balance cannot realistically be achieved. Careful monitoring and stringent maintenance practices must be observed to ensure proper function and longevity of the system. The original design should take into account the type of fluid intended for use in the system.
Any hydraulic system that uses a conductive type of water-based fluid will, no doubt, be subject to enhanced corrosion potential. To avoid this problem:
- Select components carefully to avoid using those made of dissimilar metals.
- Carefully monitor any DC electrical functions incorporated in the system to locate and resolve any poor grounding or other source of leaking voltage.
- The fluid is the single most important component in any hydraulic system, so regularly analyze it to monitor the overall health of the system.
- Consider using additives to decrease the electrical conductivity of the fluid.